 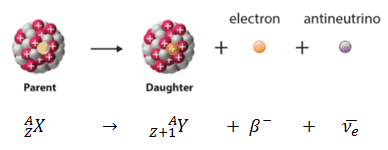
There is one kind of double beta decay – called neutrino-less double beta decay (the image above is from the COBRA Project, one study of this) – which is being studied intensely (though no such decay has yet been observed), because it may be one of the very few easily opened windows into physics beyond the Standard Model (see this WIPP page for more details). There is also a rare double beta decay, in which two beta particles are emitted it has been observed, in some unstable isotopes, as predicted. In nuclear physics, beta decay is a type of radioactive decay in which a beta particle (an electron or a positron) is emitted. In beta minus ( ) decay, a neutron is converted to a proton, and the process creates an electron and an electron antineutrino while in beta plus ( +) decay, a proton is converted to a neutron and the process creates a positron and an electron neutrino. 2 <2.2 × 106MeV, we can safely ignore the neutrino rest mass energy, within the experimental uncertain of the reaction Q, 4 CHAPTER 15. The two types of beta decay are known as beta minus and beta plus. So why do isolated neutrons decay (but those in stable nuclei, and those in neutron stars, don’t)? And why are isolated protons stable, but those in certain radioactive nuclei not? It’s all down to energy … if one state (an isolated neutron, say) has a higher energy than another (proton plus electron plus antineutrino), then the first will decay into the second (the baryon number of the two states must be the same, ditto lepton number, and so on). Consequences of -decay’s 3-body nal state. In beta minus decay, a neutron changes into a proton, antineutrino, and electron this conversion is due to the weak interaction (or weak force) … a down quark (in the neutron) becomes an up quark and emits a W – boson (one of three bosons which mediate the weak interaction), which then decays into an electron and an antineutrino.īeta plus decay – which is also known as inverse beta decay – involves the conversion of a proton to a neutron, positron, and neutrino. It was also in 1934 that Fermi published – in Italian and German (Nature considered the idea too speculative!!) – his theory of beta decay (for more details on this, check out this Hyperphysics page). It is accompanied by the emission of an electron antineutrino which shares the momentum and energy of. 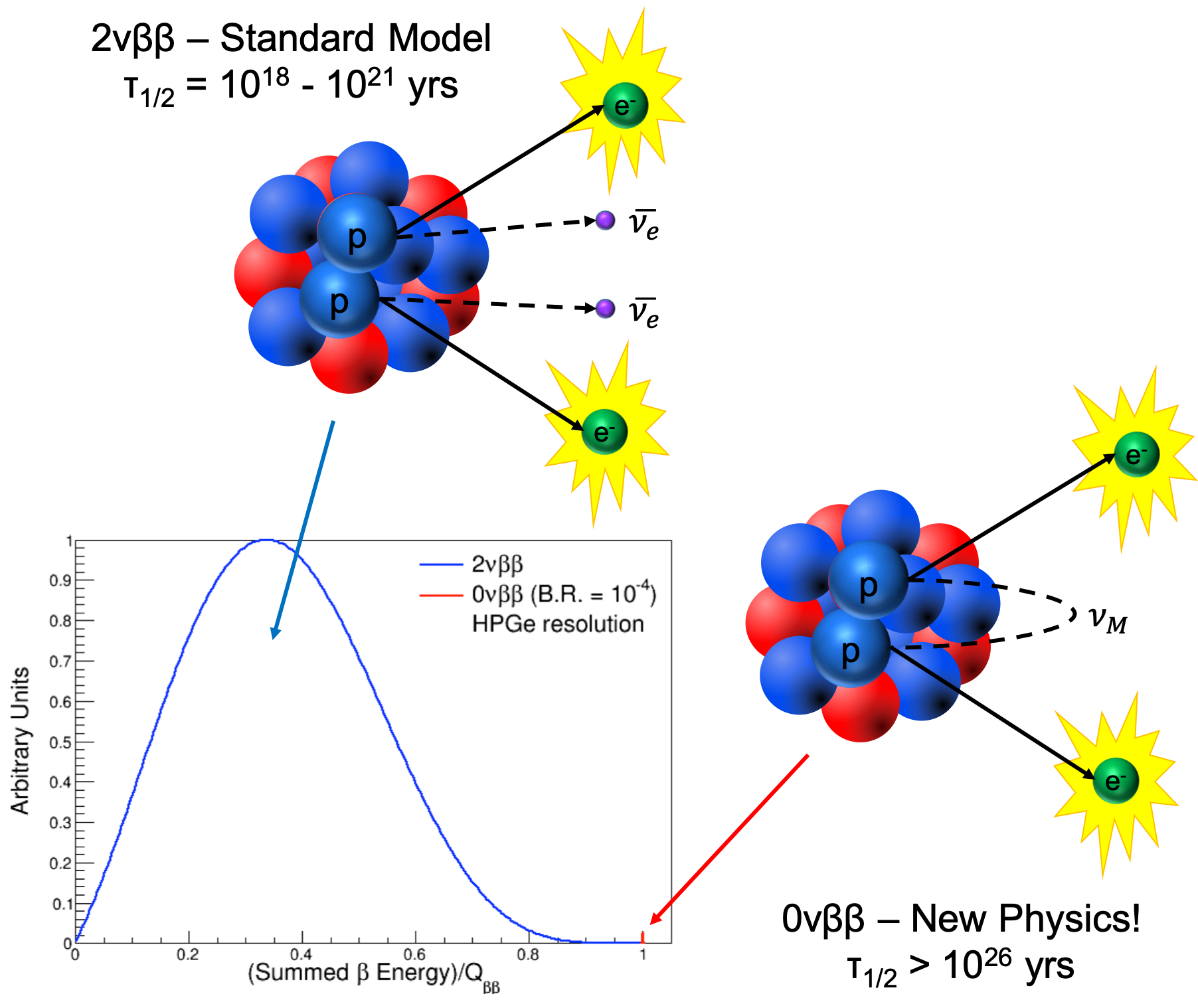 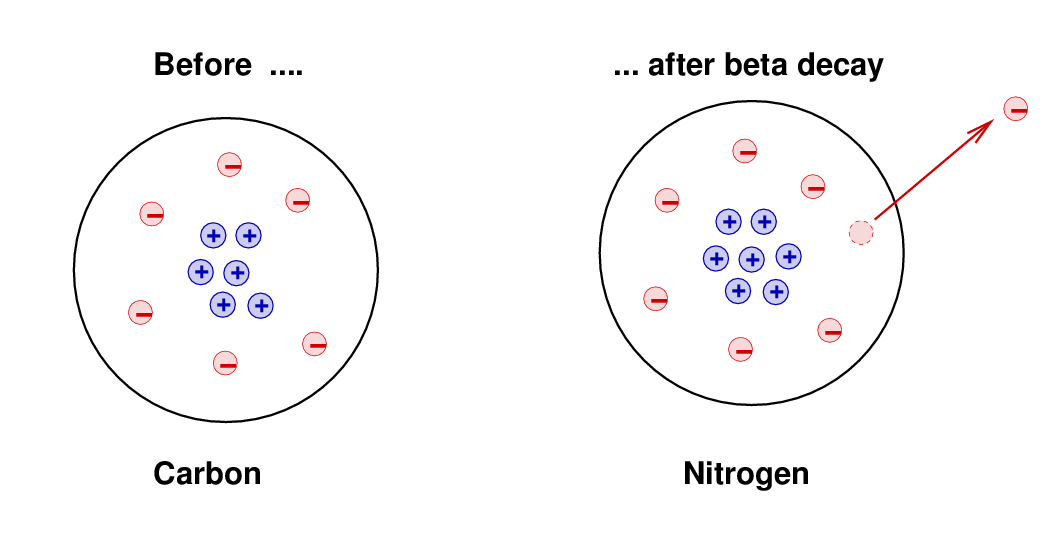
However, this is beta minus decay … the discovery of beta plus decay (by Irène and Frédéric Joliot-Curie, in 1934) came after the discovery of the positron (in cosmic rays, in 1932) and the (then) controversial ‘invention’ of the neutrino (by Pauli, in 1931) to account for the continuous energy spectrum of electrons in beta decay. Beta emission has a characteristic energy spectrum. Beta decay is when an unstable atomic nucleus decays (radioactively) by emitting a beta particle when the beta particle is an electron, it is β – decay, and when a positron, β + decay.īeta rays, as a distinct component of the rays given off in radioactivity, were discovered by Rutherford, in 1899, just a few years after radioactivity itself was discovered (in 1896). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
